There are only five atoms that will appear in your amino acid variable groups: H, C, N, O, and S. The lack of polarity means they have no way to interact with highly polar water molecules, making them water fearing. Exploring the role of hydrophilic amino acids in unfolding of protein in aqueous ethanol solution. Hydrophobic amino acids have little or no polarity in their side chains. Alanine and Valine, for example, are both nonpolar amino acids. Key words: sorption, amino acids, activated carbon, alanine, arginine, phenylalanine. The difference between a polar (water) and nonpolar (ethane) molecule is due to the. It was found that sorption of amino acids depends on the chemistry of the sorbent surface, the pH of the equilibrium solution and the structure of the amino acid molecule. How do you know if an amino acid is hydrophobic? amino acids with synthetic carbonaceous sorbents from aqueous solutions was studied. From such plots it is possible to predict what proportion of the proteins that an organism makes are transmembrane proteins.

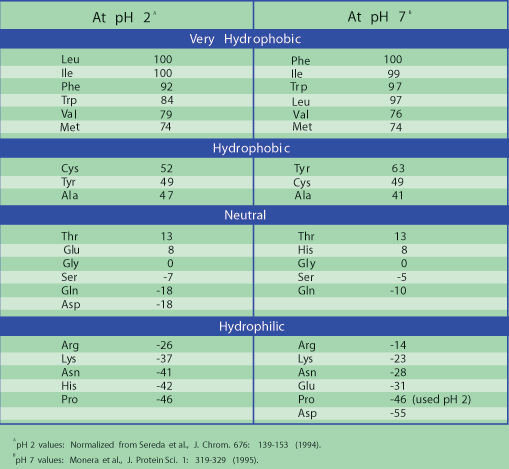
Polar amino acid residues have a tendency to be on the outside of a protein, due to the hydrophilic properties of the side chain. Segments containing about 2030 amino acids with a high degree of hydrophobicity are long enough to span a lipid bilayer as an helix, and they can often be identified by means of a hydropathy plot (Figure 10-20). Are all polar amino acids hydrophilic?Īll polar amino acids have either an OH or NH2 group (when in aqueous environment), and can therefore make hydrogen bonds with other suitable groups. Which amino acid would you expect to find in the core of a protein that is in a solution of water Possible Answers: Threonine. Where are hydrophilic amino acids found?īecause the charged and polar amino acids are hydrophilic, they are usually found at the surface of a water-soluble protein, where they not only contribute to the solubility of the protein in water but also form binding sites for charged molecules. This use of experimental X-ray scattering data provides an alternative approach for testing amino acid interactions in aqueous solution that has mostly relied on osmotic pressure measurements in the past.3134 There has also been a systematic study by Elcock and colleagues to derive eective potentials from atomistic MD simulations.35. The hydrophobic amino acids include alanine (Ala, A), valine (Val, V), leucine (Leu, L), isoleucine (Ile, I), proline (Pro, P), phenylalanine (Phe, F) and cysteine (Cys). For discussion of OH−π, and CH−O types of hydrogen bonds see Scheiner et al., 2002.
